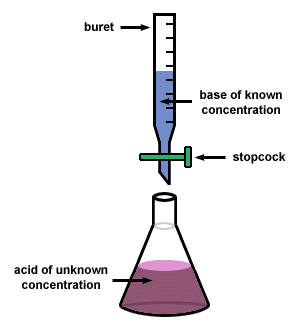
Titration Definition:
A titration with an acid and base is a neutralization reaction that determines what the unknown concentration is.
Materials:
1. Any Acid
2. Any Base
3. A buret
4. Indicator
5. 3 Beakers, one for the acid, one for the base and one for waste
6. Conical Flask
7. Volumetric pipet with a pump
8. Funnel
Procedure:
1. Fill the acid beaker with the acid
2. Fill the base beaker with the base
3. Pour the acid into the buret
4. Extract 10 cc of the base using the volumetric pipet
5. Pour the 10 cc of base into a conical flask
6. Drop 3 droplets of the indicator into the 10 cc of base
7. Put the conical flask under the buret
8. Pour the acid in the buret into the conical flask
9. Stir the conical flask while the acid is being poured into the conical flask
10. Stop pouring once the solution turns clear again
11. Wright down the difference between when you started pouring and when you finished
12. The difference is your answer
A titration with an acid and base is a neutralization reaction that determines what the unknown concentration is.
Materials:
1. Any Acid
2. Any Base
3. A buret
4. Indicator
5. 3 Beakers, one for the acid, one for the base and one for waste
6. Conical Flask
7. Volumetric pipet with a pump
8. Funnel
Procedure:
1. Fill the acid beaker with the acid
2. Fill the base beaker with the base
3. Pour the acid into the buret
4. Extract 10 cc of the base using the volumetric pipet
5. Pour the 10 cc of base into a conical flask
6. Drop 3 droplets of the indicator into the 10 cc of base
7. Put the conical flask under the buret
8. Pour the acid in the buret into the conical flask
9. Stir the conical flask while the acid is being poured into the conical flask
10. Stop pouring once the solution turns clear again
11. Wright down the difference between when you started pouring and when you finished
12. The difference is your answer

